The pharmaceutical and biotechnology industries are increasingly reliant on robust and standardized data collection and reporting. Accurate and complete case reports are fundamental to ensuring the integrity of clinical trials, facilitating informed decision-making, and ultimately, advancing medical research and patient care. A well-structured case report form template is therefore a critical tool for streamlining this process. This article will delve into the importance of these templates, exploring their key components, benefits, and practical considerations for researchers, clinicians, and regulatory bodies. The core focus will be on understanding how to effectively utilize a case report form template to ensure compliance and maximize the value of clinical trial data. Case Report Form Template Clinical Trials is more than just a document; it’s a strategic asset for optimizing clinical trial operations.
The evolution of clinical trials has dramatically increased the complexity of data collection. Traditional methods often lacked the granularity and standardization needed to effectively track patient outcomes, adverse events, and treatment responses. The rise of electronic data capture (EDC) systems and the increasing volume of data have further amplified these challenges. A standardized case report form template provides a consistent framework for capturing and organizing this data, reducing errors, and improving the efficiency of the trial process. It’s a shift from fragmented data to a more holistic and actionable record. The ability to quickly access and analyze data from a standardized template is a significant advantage in a competitive research landscape. Furthermore, adherence to established templates promotes transparency and accountability, bolstering trust within the scientific community. Ultimately, investing in a robust case report form template is an investment in the quality and reliability of clinical trial results.
Understanding the Importance of Case Report Form Templates
Before diving into the specifics of template design, it’s crucial to understand why these templates are so vital. They address several key challenges within clinical trials:
- Data Standardization: Templates ensure that data is collected and recorded in a consistent format, minimizing variability and improving comparability across different trials.
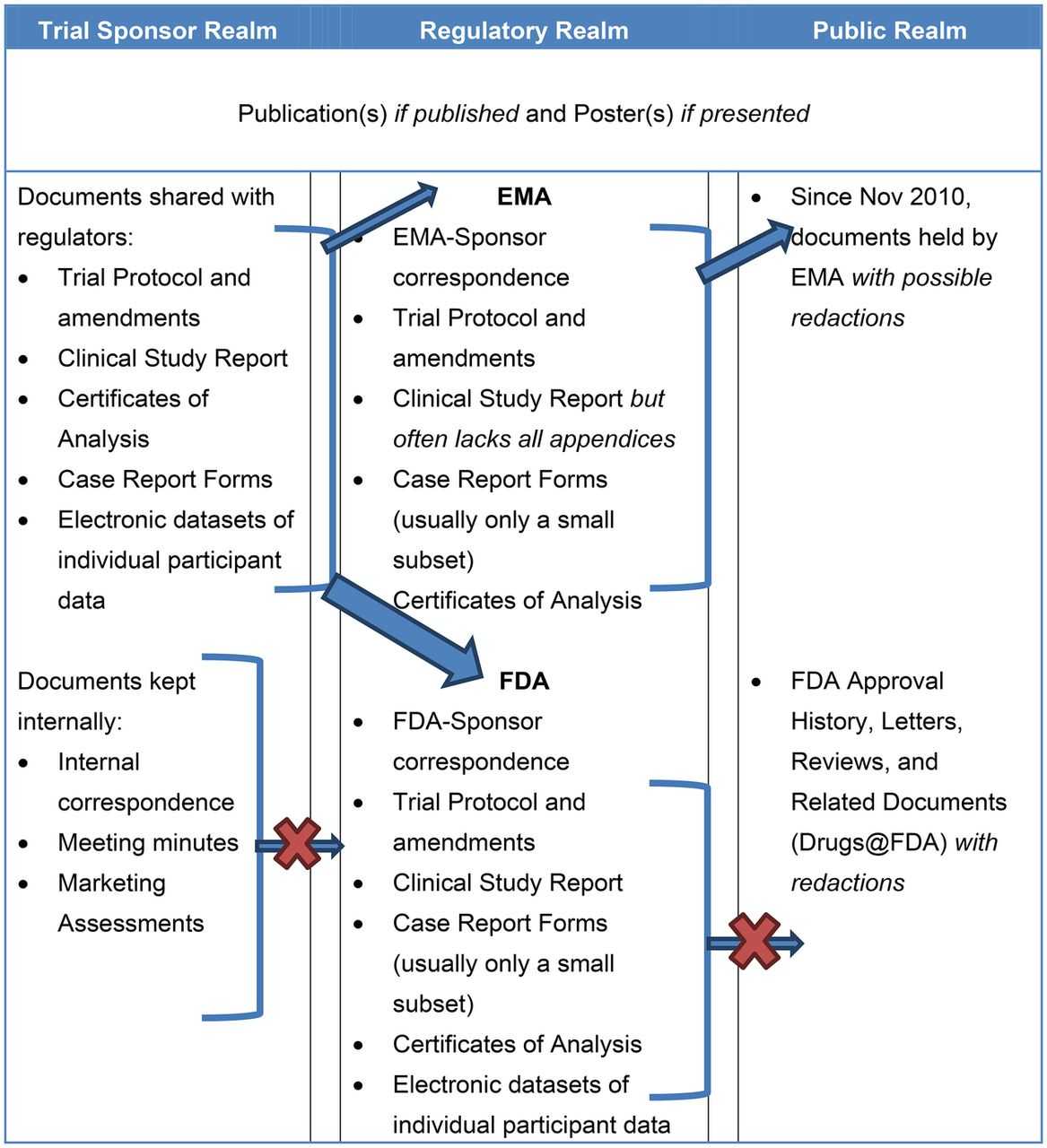
- Regulatory Compliance: Regulatory agencies like the FDA and EMA require detailed and comprehensive case reports. Using a standardized template facilitates compliance with these requirements.
- Improved Data Quality: Structured data collection minimizes errors and inconsistencies, leading to higher-quality data.
- Streamlined Reporting: Templates simplify the reporting process, reducing the time and effort required to produce reports.
- Enhanced Collaboration: A shared template allows for seamless data exchange and collaboration among researchers, clinicians, and regulatory staff.
Key Components of a Case Report Form Template
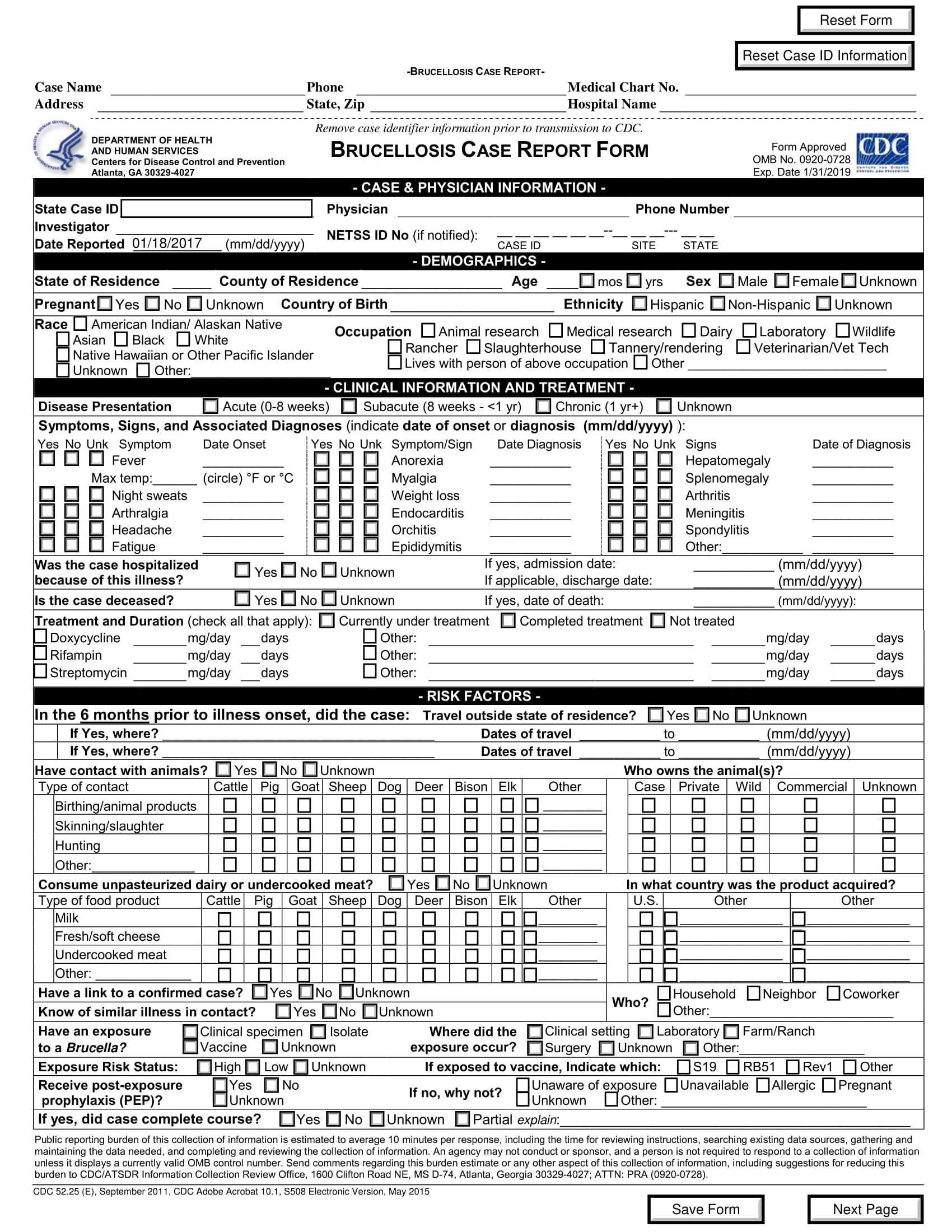
A comprehensive case report form template typically includes several key sections, each designed to gather specific information relevant to the trial. Here’s a breakdown of the essential elements:
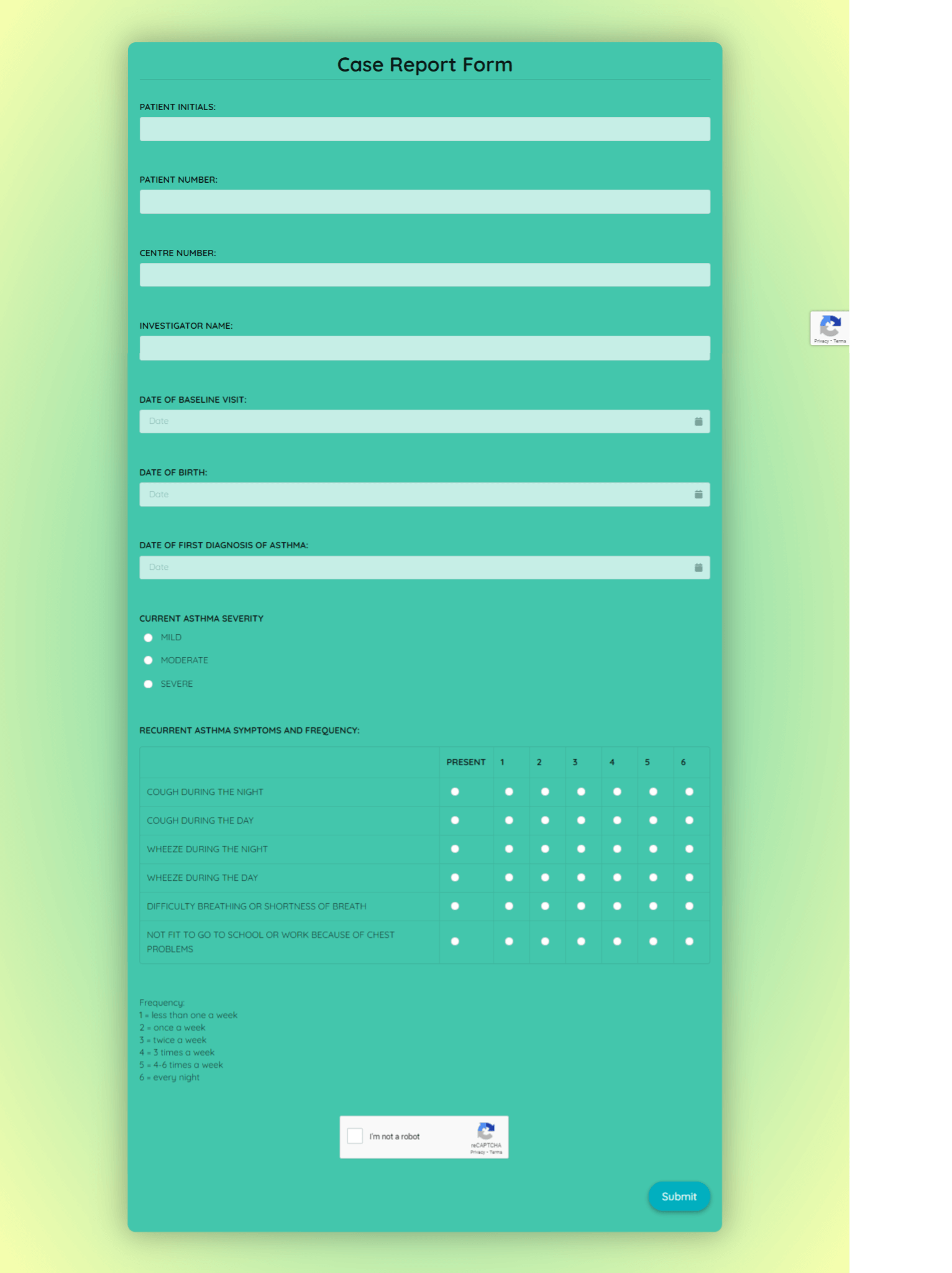
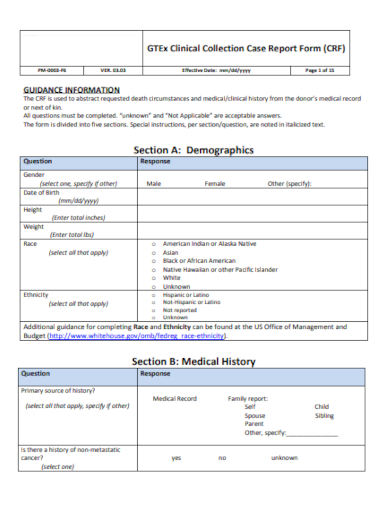
1. Patient Demographics
This section gathers basic information about the study participant, including:
- Patient ID: A unique identifier for each patient.
- Age: Patient’s age at enrollment.
- Gender: Patient’s gender.
- Ethnicity: Relevant ethnic background information.
- Medical History: A summary of the patient’s relevant medical history, including pre-existing conditions and medications.
- Contact Information: Patient’s contact details for follow-up communication.
2. Study Information
This section provides context about the trial itself:
- Trial Title and Number: The official name and identifier of the clinical trial.
- Study Design: Description of the trial design (e.g., randomized controlled trial, observational study).
- Study Objectives: Clearly stated goals of the trial.
- Study Population: Detailed description of the inclusion and exclusion criteria for participants.
- Start and End Dates: The timeline of the trial.
3. Intervention Details
This is arguably the most critical section, detailing the treatment or intervention being studied:
- Treatment Protocol: A comprehensive description of the treatment regimen, including dosage, frequency, and route of administration.
- Drug Name and Dosage: Specific details about the medication being administered.
- Alternative Treatments: If applicable, a description of any alternative treatments used or considered.
- Study Drug Name: The name of the drug being administered.
4. Outcome Measures
This section outlines the specific outcomes being measured:
- Primary Outcome: The main outcome measure of interest.
- Secondary Outcomes: Additional outcomes measured to provide a more complete picture of the treatment effect.
- Measurement Methods: How the outcome measures will be assessed (e.g., blood tests, imaging, questionnaires).
- Data Collection Schedule: When and how data will be collected for each outcome measure.
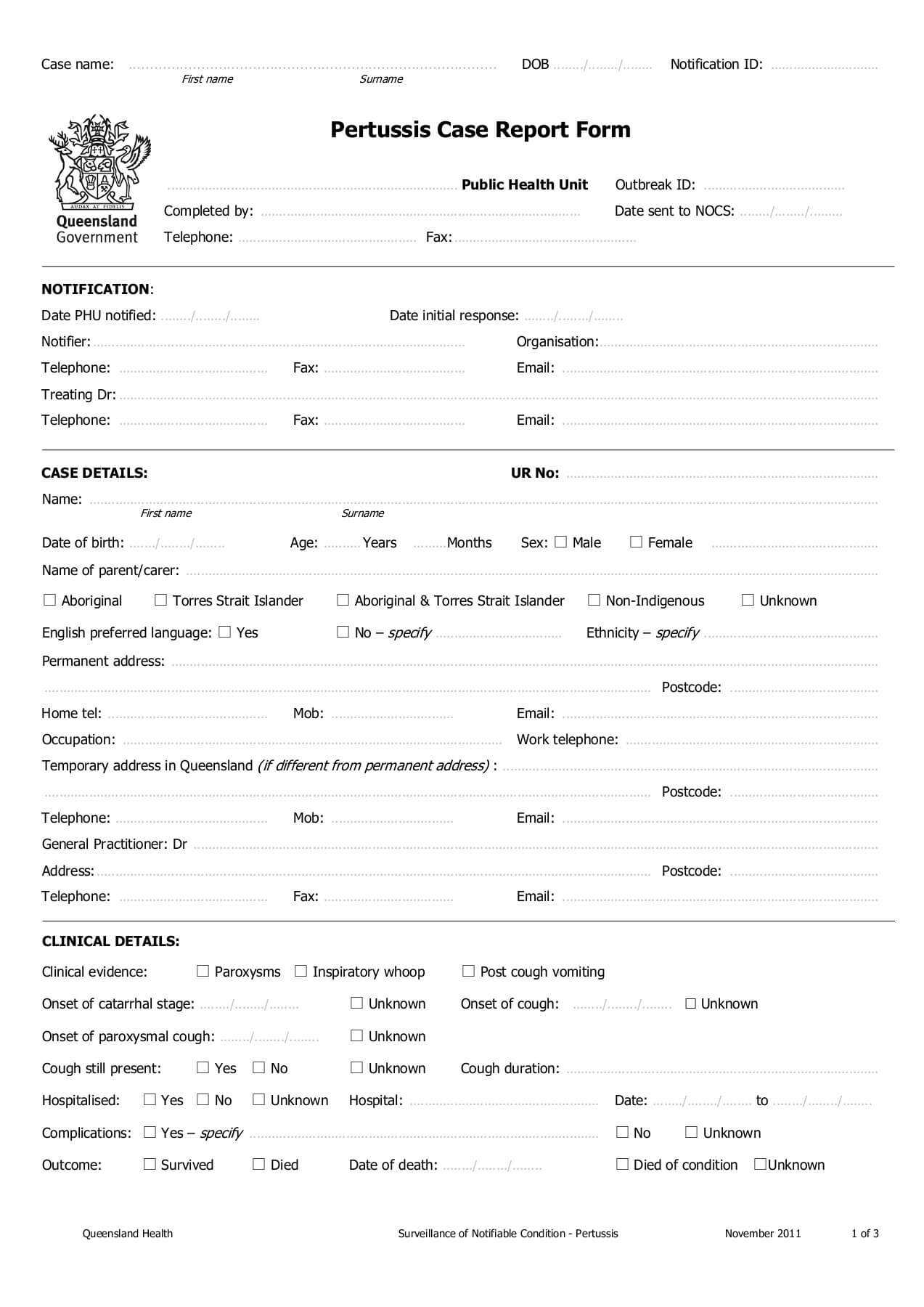
5. Adverse Events
This section documents any adverse events (AEs) that occur during the study:
- AE Description: A detailed description of the adverse event, including onset, severity, and any related symptoms.
- AE Severity: Assessment of the severity of the AE (e.g., mild, moderate, severe).
- Treatment Response: Information about how the AE was managed (e.g., supportive care, medication).
- Reporting Procedures: Instructions on how to report AEs to the sponsor.
6. Study Procedures
This section provides a detailed account of the procedures performed during the trial:
- Informed Consent: A record of the informed consent process.
- Baseline Data: Data collected before the start of the study.
- Follow-up Data: Data collected during the study period.
- Laboratory Tests: Results of laboratory tests performed during the study.
Benefits of Utilizing a Case Report Form Template
Implementing a well-designed case report template offers a multitude of advantages:
- Reduced Errors: Standardized data collection minimizes human error.
- Improved Data Quality: Consistent data capture leads to more reliable results.
- Increased Efficiency: Streamlined reporting processes save time and resources.
- Enhanced Collaboration: A shared template facilitates seamless data exchange.
- Regulatory Compliance: Template adherence ensures compliance with regulatory requirements.
- Faster Analysis: Easier access to comprehensive data enables quicker analysis and interpretation.
Best Practices for Template Design
Creating an effective case report form template requires careful consideration of several factors:
- Clarity and Simplicity: Use clear, concise language and avoid jargon.
- Completeness: Ensure all required sections are included.
- Consistency: Maintain consistent formatting and terminology throughout the template.
- Accessibility: Design the template to be easily accessible and navigable.
- User-Friendliness: Consider the needs of the intended users when designing the template.
The Role of Technology in Case Report Form Templates
Modern technology is playing an increasingly important role in the design and implementation of case report templates. Electronic data capture (EDC) systems, data management software, and cloud-based platforms are transforming the way clinical trial data is collected and managed. These technologies offer several benefits, including:
- Automated Data Entry: EDC systems can automatically populate data fields, reducing manual data entry errors.
- Real-Time Data Validation: Software can validate data as it is entered, ensuring accuracy.
- Secure Data Storage: Cloud-based platforms provide secure storage and access to data.
- Data Analysis Tools: Software can facilitate data analysis and reporting.
Challenges and Considerations
While templates offer significant benefits, there are also challenges to consider:
- Template Customization: Adapting templates to specific trial designs and data requirements can be time-consuming.
- Training: Researchers and staff need to be trained on how to use the template effectively.
- Data Security: Protecting patient data is paramount, and templates must be designed with security in mind.
- Maintaining Compliance: Templates must be regularly updated to reflect changes in regulatory requirements.
Conclusion
Case report form templates are an indispensable tool for modern clinical trials. By providing a standardized framework for data collection and reporting, these templates significantly improve data quality, streamline the trial process, and enhance regulatory compliance. Investing in a robust and well-designed template is a strategic investment in the success of clinical trials and the advancement of medical research. The continued evolution of EDC and cloud-based technologies will undoubtedly further enhance the capabilities and usability of these templates, solidifying their position as a cornerstone of modern clinical trial operations. Case Report Form Template Clinical Trials remains a critical component of a successful clinical trial strategy.